Quantum chemical calculations are applied to study the (CT) complexes in order to obtain information on structures and other molecular properties like specific interaction of donor and acceptor.
#HOMO LUMO ORGANIC CHEMISTRY FREE#
Quantum chemical calculation of geometrical structure and energies were studied using PM3 and AM1 methods in where the Gibbs free energy was calculated with a value of -9028 and -5796 Kcal/mol, respectively these values showed that the reaction was carried out.
#HOMO LUMO ORGANIC CHEMISTRY SOFTWARE#
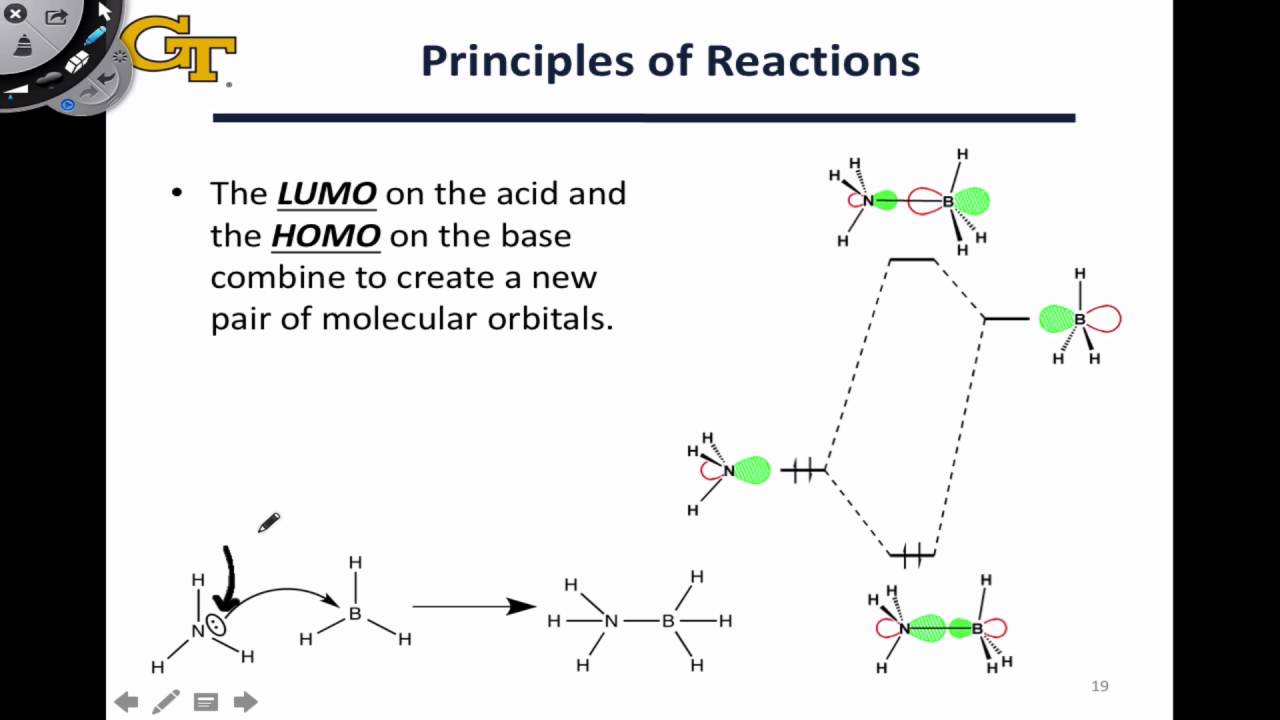
Centro, Hermosillo, Sonora, MéxicoĪbstract Chitosan and PVP oligomers were analyzed by means of the HyperChem software 8.0v to determine the theoretical structure. ‡ Departamento de Investigación y Posgrado en Alimentos. 20256 Aguascalientes, Aguascalientes, México. † División de Estudios de Posgrado e Investigación del Instituto Tecnológico de Aguascalientes, Ave. Top: σ interaction.Analysis of chitosan/polyvinylpyrrolidone (structure, ftir, electrostatic potential, homo/lumo orbitals) using computational chemistry MO interactions between Fe and CO frontier MOs. And because the HOMO and LUMO of CO are big on carbon, you won't be surprised that the bond is Fe-C=O, not Fe-O=C. This "multiple bond" between CO and Fe(II) makes CO toxic. So what can happen is that the HOMO on CO makes a σ bond with the LUMO on Fe(II), and the HOMO on Fe(II) makes a π bond with the LUMO on CO. The HOMO is high, because 3d is not so stable, and the LUMO is low, because it is also 3d and not much higher than the HOMO (there is a gap because the other atoms around the Fe(II) in hemoglobin make the d orbitals different energies). Fe(II) also has a small gap between HOMO and LUMO, because it has 6 electrons in 3d orbitals. CO has a small gap between the HOMO and LUMO. The LUMO is a π* orbital that is also mostly on carbon, and it is kind of low-energy because the splitting of π MOs is smaller than σ MOs (because of less overlap). The HOMO is a slightly-bonding orbital that is mostly on carbon. Go back and look at the MO diagram for CO. We can understand how and why CO binds Fe(II) using MO theory. CO binds to the Fe(II) more tightly than O 2, so if you breathe too much CO, your cells won't get any oxygen, because all the Fe(II) in your blood bound CO instead. You use Fe(II) ions in your blood to bind O 2 and carry it to your cells. The reason CO is toxic is because it binds metal ions really tightly. You probably know that CO is toxic (which is why you shouldn't stay in a garage with a car running, because the CO from incomplete combustion can kill you). This is why if you want to store something reactive like K metal, you probably keep it in a bottle of oil. They do burn easily, but you have to get them hot first. Molecules with low HOMO and high LUMO, a big HOMO-LUMO gap, are not very reactive! Hydrocarbons are a good example (like oil, etc). Diagrams of acid/base, redox, and both reactions. You can still predict covalent/ionic bonding based on electronegativity. This depends on the AO energies, which depends on the electronegativity, just like you would expect. Often the energy match is bad, so that no covalent bond forms, just the electron moves. In a redox reaction, the oxidant has a low LUMO, and the reductant has a high HOMO, but this time the oxidant LUMO is lower than the reductant HOMO, so that the electrons in the reductant HOMO move completely to the oxidant LUMO. This creates a bond between the acid and base. A low LUMO usually means an empty valence orbital, like on B or on a cation.) We can make a bonding and anti-bonding combination of the base HOMO and acid LUMO, and that will stabilize the electrons from the base's HOMO, lowering the total energy. (A lone pair in MO theory is an electron pair in a non-bonding MO. What this really means is that the base has a HOMO that is pretty high-energy (a lone pair), and the acid has a LUMO that is pretty low energy. In a Lewis acid/base reaction, an electron pair from the base is shared with the acid. Earlier, we said that most reactions can be called either Lewis acid/base or redox.
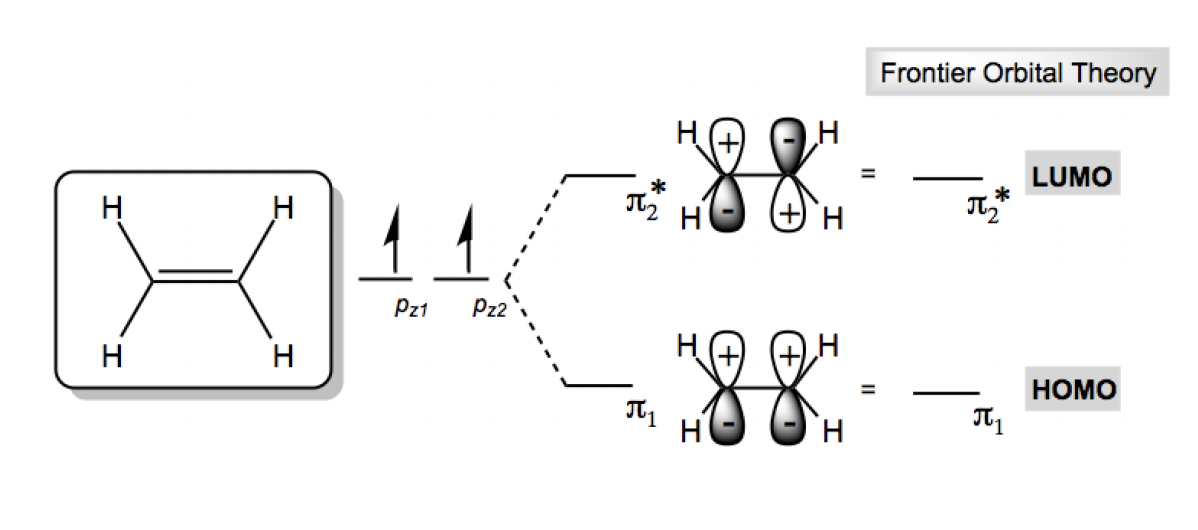
LUMO is Lowest Unoccupied MO, the lowest-energy MO that doesn't have any electrons in it.įrontier MOs are very important for reactivity. HOMO is Highest Occupied MO, the highest-energy MO that has electrons in it. The frontier MOs are called the HOMO and the LUMO. In this case, we are interested in the MOs at the border between occupied and empty. Distinguish and describe the significance of frontier MOsįrontier means a border area, between two things (often, between 2 countries).
